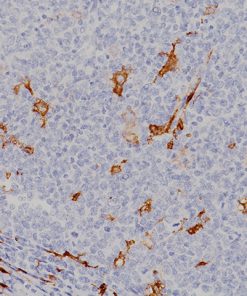
Microphthalmia Transcription Factor (MiTF)
$350.00 – $1,110.00Price range: $350.00 through $1,110.00
Description
Product Description
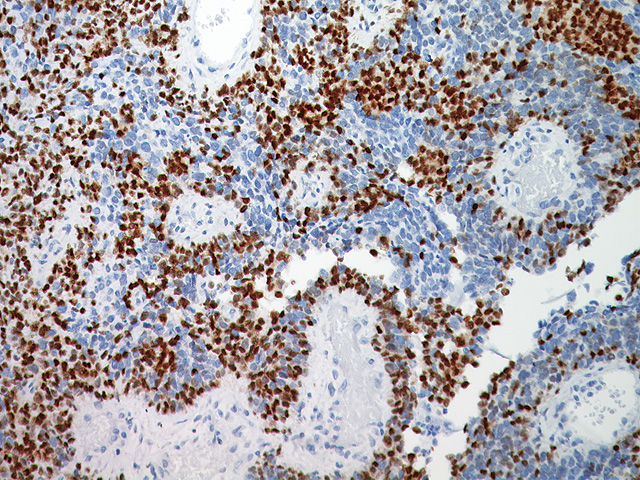
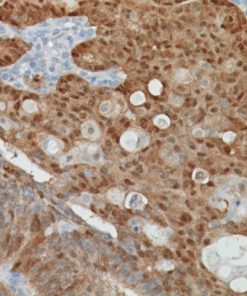
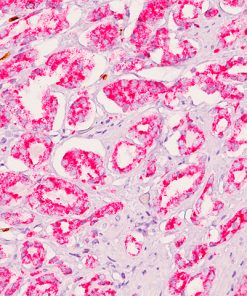
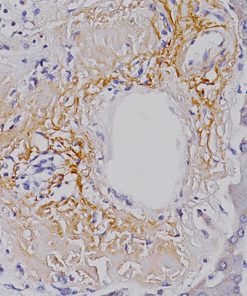
Microphthalmia Transcription Factor antibody (MiTF) is a nuclear melanocytic marker. Studies have shown it is a sensitive and specific marker for malignant melanoma, including some spindle-cell variants. MiTF has been shown to have superior sensitivity and specificity to S100 and HMB45. MiTF may be useful for identification of melanoma, melanocytic soft tissue tumors and the unusual group of tumors that show combined melanocytic and myloid differentiation, the perivascular epithelioid cell family of tumors (PEComas). Microphthalmia Transcription Factor may be a valuable addition to a melanoma marker panel with S-100, HMB45, Tyrosinase and MART-1.
Specifications
Specifications
| Intended Use | |
|---|---|
| Species Reactivity | |
| Source | |
| Clone | |
| Isotype | |
| Antigen | |
| Localization | |
| Positive Control |
Datasheets & SDS
| Download Data Sheet |
| Download ONCORE Pro Data Sheet |
| Download SDS Sheet |
Browse more documents for this product (IFUs, datasheets, translations, SDS, and more).
References
1. Ohsie SJ, et al. Immunohistochemical characteristics of melanoma. J Cutan Pathol. 2008 May; 35(5):433-44.
2. Sheffield MV, et al. Comparison of five antibodies as markers in the diagnosis of melanoma in cytologic preparations. Am J Clin Pathol. 2002 Dec; 118(6):930-6.
3. Dorvault CC, et al. Microphthalmia transcription factor: a sensitive and specific marker for malignant melanoma in cytologic specimens. Cancer. 2001 Oct 25; 93 (5):337-43.
4. O’Reilly FM, et al. Microphthalmia transcription factor immunohistochemistry: a useful diagnostic marker in the diagnosis and detection of cutaneous melanoma, sentinel lymph node metastases, and extracutaneous melanocytic neoplasms. J Am Acad Dermatol. 2001 Sep; 45(3):414-9.
5. Miettinen M, et al. Microphthalmia transcription factor in the immunohistochemical diagnosis of metastatic melanoma: comparison with four other melanoma markers. Am J Surg Pathol. 2001 Feb; 25(2):205-11.
6. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
7. National Committee for Clinical Laboratory Standards (NCCLS). Protection of laboratory workers from infectious diseases transmitted by blood and tissue; proposed guideline. Villanova, PA 1991;7(9). Order code M29-P.