Mart-1 Cocktail
$295.00 – $2,306.00Price range: $295.00 through $2,306.00
Description
Product Description
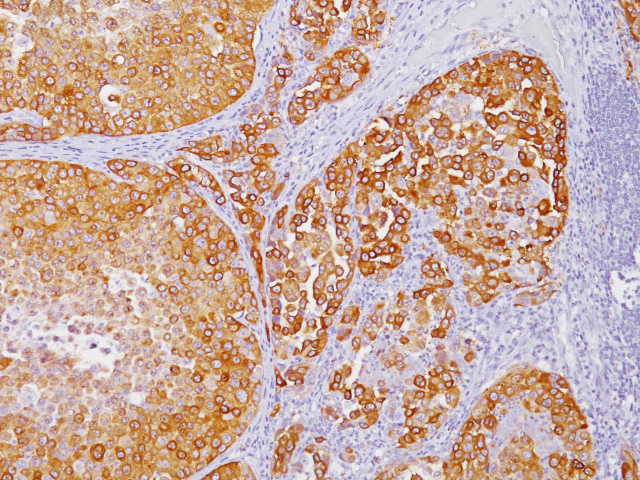
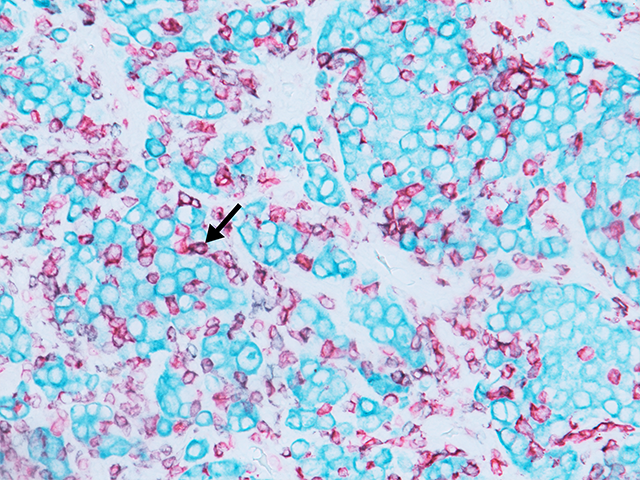
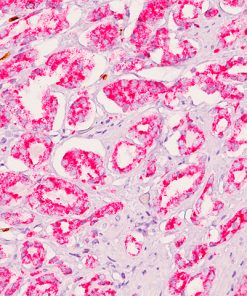
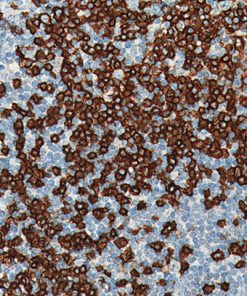
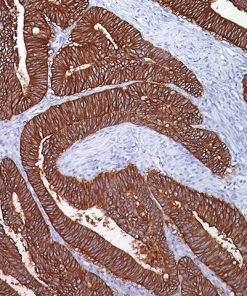
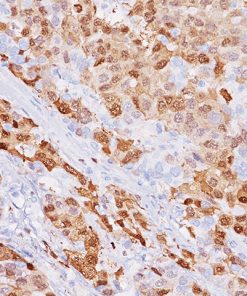
The MART-1/Melan A recognizes a protein of 18 kDa, identified as MART-1 (Melanoma Antigen Recognized by T cells 1) or Melan-A. The MART-1 recognizes a subcellular fraction found in melanosomes. The antibody labels melanomas and tumors showing melanocytic differentiation. It does not mark neoplasms of epithelial origin, lymphomas or mesenchymal tumors. MART-1 is a useful addition to melanoma panels which are specific to melanocytic lesions. Both HMB-45 and MART-1 are coexpressed in the majority of melanomas, as well as solely expressed in certain cases. Studies have shown that MART-1 is more sensitive than HMB-45 when labeling metastatic melanomas.15-17
Specifications
Specifications
| Weight | N/A |
|---|---|
| Dimensions | N/A |
| Intended Use | |
| Species Reactivity | |
| Source | |
| Clone | |
| Isotype | |
| Antigen | |
| Localization | |
| Positive Control |
Datasheets & SDS
| Download CE Marked IVD Datasheet |
| Download IVD Datasheet Without CE Mark |
| Download Q Series Data Sheet |
| Download RUO Data Sheet for International |
| Download SDS Sheet |
Browse more documents for this product (IFUs, datasheets, translations, SDS, and more).
Regulatory Notice: Biocare’s IVD-labeled products comply with US-FDA and European IVDD regulation. Other regions may have additional requirements for such labeling, please contact your local distributor.
Oncore Protocol
| Antibody | Download |
|---|---|
| Download Oncore Protocol |
References
1. Orchard GE. Melan A (MART-1): a new monoclonal antibody for malignant
melanoma diagnosis. Br J Biomed Sci 1998 Mar; 55(1):9-9.
2. Blessing K, Sanders DS, Grant JJ. Comparison of immunohistochemical staining of
the novel antibody Melan-A with S100 protein and HMB-45 in malignant melanoma
and melanoma variants. Histopathology 1998 Feb; 32(2):139-146.
3. Kageshita T, et al. Differential expression of MART-1 in primary and metastatic
melanoma lesions. J Immunother 1997 Nov; 20(6):460-465.
4. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22,
Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove
Azide Salts.”
5. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory
workers from occupationally Acquired Infections; Approved guideline-Third Edition
CLSI document M29-A3 Wayne, PA 2005.
Related products
25ml
25ml
25ml