Description
Product Description
Tyrosinase is a key enzyme involved in the initial stages of melanin biosynthesis. Studies have shown Tyrosinase antibody to be a more sensitive marker for melanoma when compared to HMB45 and MART-1. It has also shown to label a higher percentage of desmoplastic melanomas than HMB45. Unlike HMB45, Tyrosinase does not discriminate between activated or resting melanocytes. Other studies have shown Tyrosinase to be a very specific marker for melanomas that did not cross react with any tumors or normal tissues tested. Tyrosinase is reported to be a superior melanoma marker when compared to HMB45.
Specifications
Specifications
| Intended Use | |
|---|---|
| Species Reactivity | |
| Source | |
| Clone | |
| Isotype | |
| Antigen | |
| Localization | |
| Positive Control |
References
1. Orchard GE. Comparison of immunohistochemical labelling of melanocyte differentiation antibodies melan-A, tyrosinase and HMB 45 with NKIC3 and S100 protein in the evaluation of benign naevi and malignant melanoma. Histochem J 2000 Aug;32(8):475-81.
2. Jungbluth AA, Iversen K, Coplan K, Kolb D, Stockert E, Chen YT, Old LJ, Busam K. T311–an anti-tyrosinase monoclonal antibody for the detection of melanocytic lesions in paraffin embedded tissues. Pathol Res Pract 2000;196(4):235-42.
3. Kaufmann O, Koch S, Burghardt J, Audring H, Dietel M. Tyrosinase, melan-A, and KBA62 as markers for the immunohistochemical identification of metastatic amelanotic melanomas on paraffin sections. J Cutan Pathol 1998 Apr;25(4):204-9.
4. Hofbauer GF, Kamarashev J, Geertsen R, Boni R, Dummer R. Tyrosinase immunoreactivity in formalin-fixed, paraffin-embedded primary and metastatic melanoma: frequency and distribution. J Cutan Pathol 1998 Apr;25(4):204-9
5. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
6. National Committee for Clinical Laboratory Standards (NCCLS). Protection of laboratory workers from infectious diseases transmitted by blood and tissue; proposed guideline. Villanova, PA 1991;7(9). Order code M29-P.
Datasheets & SDS
| Download Data Sheet |
| Download RUO Data Sheet for International |
| Download SDS Sheet |
Browse more documents for this product (IFUs, datasheets, translations, SDS, and more).
Regulatory Notice: Biocare’s IVD-labeled products comply with US-FDA and European IVDD regulation. Other regions may have additional requirements for such labeling, please contact your local distributor.


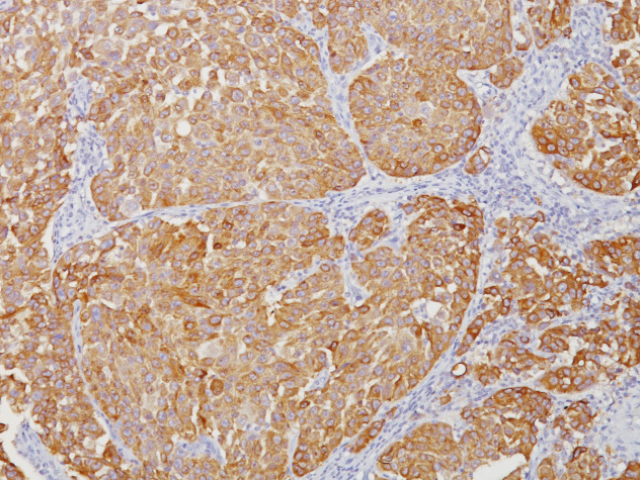
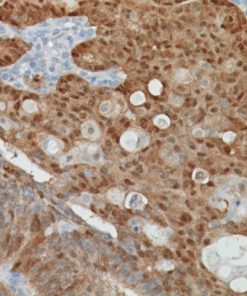
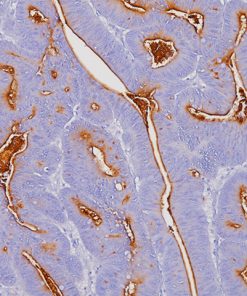
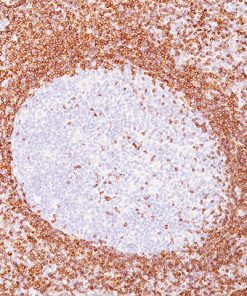
![(L) Anaplastic large cell lymphoma and (R) Lung adenocarcinoma stained with ALK antibody [5A4]](https://biocare.net/wp-content/uploads/3041-247x296.jpg)