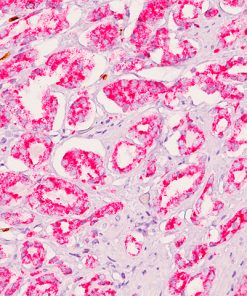
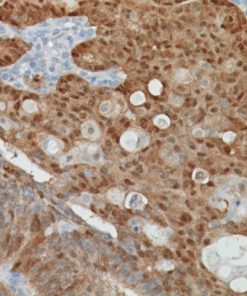
TTF-1 + Napsin A
$748.00 – $1,178.00Price range: $748.00 through $1,178.00
Description
Product Description
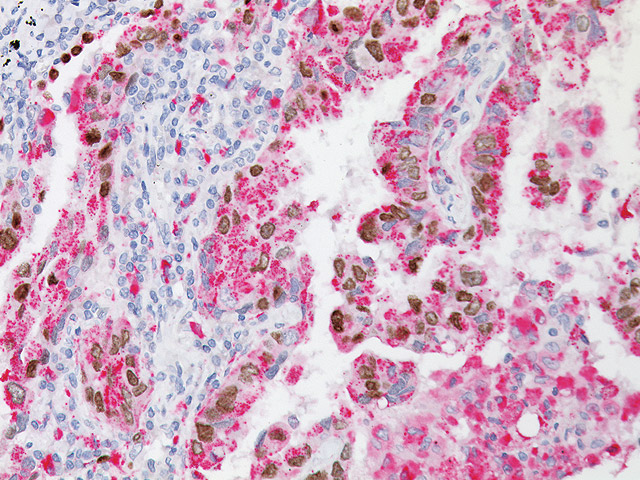
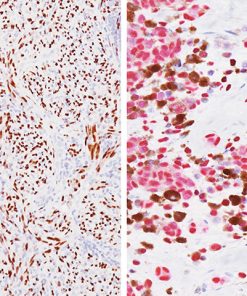
TTF-1 has been the premier marker for lung adenocarcinoma. A new and promising marker, Napsin A, is expressed in type II pneumocytes and in adenocarcinomas of the lung (2). Studies have shown Napsin A to be more sensitive and specific than TTF-1 in lung adenocarcinomas and virtually negative in all squamous carcinomas. In other studies comparing TTF-1 and SP-A, Napsin A stained more tumor cells and a higher percentage of lung adenocarcinomas than either of these antibodies (1-3). Other studies have shown that when TTF-1 and Napsin A are used in combination, a higher sensitivity and specificity is achieved (4). A critical assessment is essential for correct diagnosis as patients with squamous carcinoma (SqCC) cannot receive Avastin due to a 30% mortality rate as a result of fatal hemoptysis (hemorrhaging). Therefore, when used in a panel with p63 and CK5, this unique multiplex antibody reagent of TTF-1 and Napsin A may aid in the analysis of poorly differentiated lung adenocarcinomas vs. squamous cell carcinomas in formalin-fixed paraffin-embedded tissues.
Specifications
Specifications
| Weight | N/A |
|---|---|
| Dimensions | N/A |
| Intended Use | |
| Species Reactivity | |
| Source | |
| Clone | |
| Isotype | |
| Antigen | |
| Localization | |
| Positive Control |
Datasheets & SDS
| Download Data Sheet |
| Download RUO Data Sheet for International |
| Download SDS Sheet |
Browse more documents for this product (IFUs, datasheets, translations, SDS, and more).
Regulatory Notice: Biocare’s IVD-labeled products comply with US-FDA and European IVDD regulation. Other regions may have additional requirements for such labeling, please contact your local distributor.
References
1. Hirano T, et. al. Usefulness of TA02 (Napsin A) to distinguish primary lung adenocarcinoma from metastatic lung adenocarcinoma. Lung Cancer. 2003 Aug; 41 (2):155-62.
2. Ueno T, Linder S, Steterger G. Aspartic proteinase napsin is a useful marker for diagnosis of primary lung adenocarcinoma. Br J Cancer. 2003 Apr 22; 88(8):1229-33.
3. Suzuki A, et. al. Napsin A is useful to distinguish primary lung adenocarcinoma from adenocarcinomas of other organs. Pathol Res Pract. 2005; 201 (8-9):579-86.
4. Dejmek A, et. al. Napsin A (TA02) is a useful alternative to thyroid transcription factor-1 (TTF-1) for the identification of pulmonary adenocarcinoma cells in pleural effusions. Diagn Cytopathol. 2007 Aug; 35(8):493-7.
5. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
6. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory workers from occupationally Acquired Infections; Approved guideline-Third Edition CLSI document M29-A3 Wayne, PA 2005.
Related products
25ml
25ml