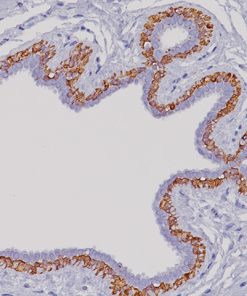
Pan Melanoma + Ki-67
$780.00 – $2,746.00Price range: $780.00 through $2,746.00
Description
Product Description
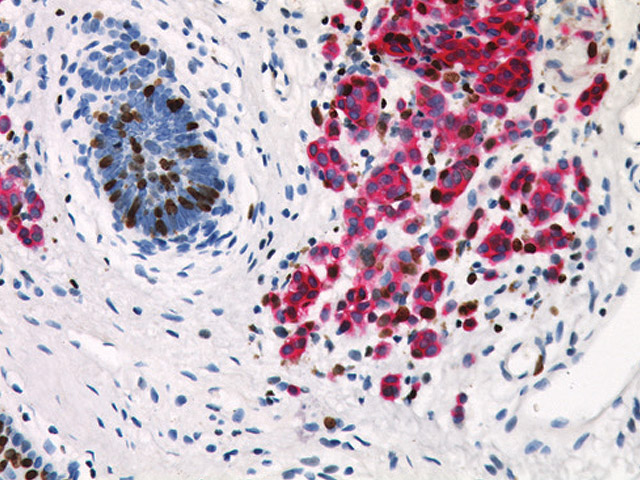
Pan Melanoma Antibody + Ki-67 serves as a tool to identify the proliferation rate of melanocytic lesions in cases in which melanocytes are sparse; there are dense lymphocytic infiltrates; and melanocytes are admixed with fibroblasts. In general, a higher proliferative fraction is seen in melanoma than in melanocytic nevi. There are many types of nevi, and some simulate melanoma closely. If the Multiplex stain shows a very low Ki-67 (DAB) labeling rate in MART-1/Tyrosinase positive cells (Fast Red), this favors benignity. A high rate, especially toward the deep part of a melanocytic lesion raises the possibility of malignancy.
Specifications
Specifications
| Weight | N/A |
|---|---|
| Dimensions | N/A |
| Intended Use | |
| Species Reactivity | |
| Source | |
| Clone | |
| Isotype | |
| Antigen | |
| Localization | MART-1 + Tyrosinase: (Cytoplasmic) Red Ki-67: (Nuclear) Brown |
| Positive Control |
Datasheets & SDS
| Download DS Data Sheet |
| Download RUO Data Sheet for International |
| Download SDS Sheet |
Browse more documents for this product (IFUs, datasheets, translations, SDS, and more).
Regulatory Notice: Biocare’s IVD-labeled products comply with US-FDA and European IVDD regulation. Other regions may have additional requirements for such labeling, please contact your local distributor.
References
1. Orchard G. Evaluation of melanocytic neoplasms: application of a pan-melanoma antibody cocktail. Br J Biomed Sci. 2002; 59(4):196-20.
2. Orchard GE. Melan A (MART-1): a new monoclonal antibody for malignant melanoma diagnosis. Br J Biomed Sci 1998 Mar; 55(1):9-9.
3. Blessing K, Sanders DS, Grant JJ. Comparison of immunohistochemical staining of the novel antibody Melan-A with S100 protein and HMB-45 in malignant melanoma and melanoma variants. Histopathology 1998 Feb; 32 (2):139-146.
4. Jansen R, et al. MIB-1 labelling index is an independent prognostic marker in primary breast cancer. Br J Cancer 1998 Aug; 78 (4):460-465.
5. Goodson WH 3rd, et al. The functional relationship between in vivo bromodeoxyuridine labeling index and Ki-67 proliferation index in human breast cancer. Breast Cancer Res Treat 1998 May; 49 (2): 155-164.
6. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
7. National Committee for Clinical Laboratory Standards (NCCLS). Protection of laboratory workers from infectious diseases transmitted by blood and tissue; proposed guideline. Villanova, PA 1991; 7(9). Order code M29-P.


![(L) Anaplastic large cell lymphoma and (R) Lung adenocarcinoma stained with ALK antibody [5A4]](https://biocare.net/wp-content/uploads/3041-247x296.jpg)