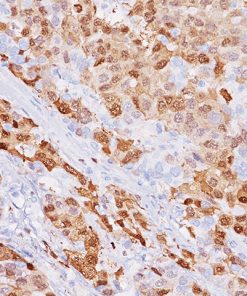
MCM2 + TOP2A
$295.00 – $3,946.00Price range: $295.00 through $3,946.00
Description
Product Description
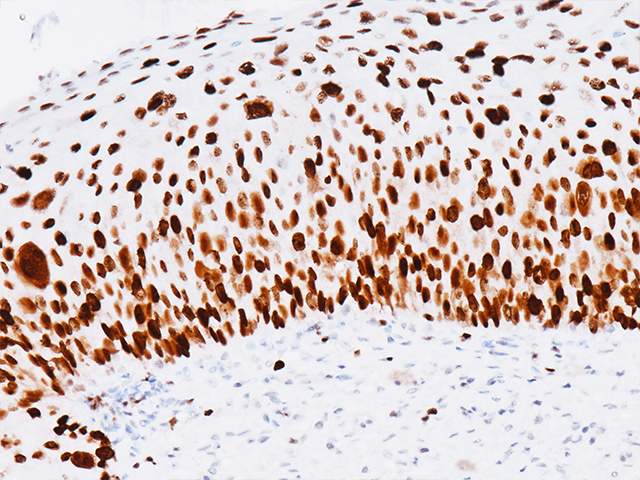
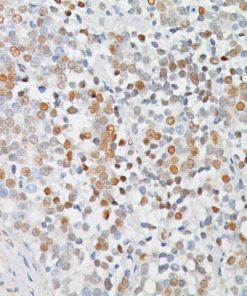
A monoclonal antibody cocktail targeted against mini-chromosome maintenance protein 2 (MCM2) and DNA topoisomerase IIA (TOP2A), when up regulated, serves as a marker of aberrant S-phase induction in proliferating cells.15 MCM2, as part of a hexameric pre-replication complex, drives the formation of pre-replicative complexes during DNA replication, a critical initial step during G1 phase.16 TOP2A is a nucleic enzyme affecting the topological structure of DNA during replication, transcription, recombination, condensation, and segregation.17,18
Specifications
Specifications
| Intended Use | |
|---|---|
| Format | |
| Volume | |
| By Letter | |
| Antigen | |
| Clone | |
| Isotype | |
| Localization | |
| Positive Control | |
| Source |
Datasheets & SDS
| Download CE Marked IVD Datasheet |
| Download IVD Datasheet Without CE Mark |
| Download RUO Datasheet |
| Download SDS Sheet |
Browse more documents for this product (IFUs, datasheets, translations, SDS, and more).
References
1. Walts AE, Bose S. p16, Ki-67, and BD ProEx™C immunostaining: a practical approach for diagnosis of cervical intraepithelial neoplasia. Hum Pathol. 2009;40:957.
2. Shi J, et al. Evaluation of p16INK4a, minichromosome maintenance protein 2, DNA topoisomerase IIA, ProEX C, and p16INK4a/ProEX C in cervical squamous intraepithelial lesions. Hum Pathol. 2007;38:1335.
3. Malinowski, DP. Molecular diagnostic assays for cervical neoplasia: emerging markers for the detection of high-grade cervical disease. Biotechniques. 2005;(Suppl 4):17.
4. Ishimi Y, et al. Enhanced expression of Mcm proteins in cancer cells derived from uterine cervix. Eur J Biochem. 2003;270:1089.
5. Wang JC. Cellular roles of DNA topoisomerases: a molecular perspective. Nat Rev Mol Cell Biol. 2002;3:430.
6. Gibbons D, et al. Comparison of topoisomerase II alpha and MIB-1 expression in uterine cervical squamous lesions. Mod Pathol. 1997;10:409.
7. Kelly D, et al. Detection of cervical high-grade squamous intraepithelial lesions from cytologic samples using a novel immunocytochemical assay (ProEx™C). Cancer. 2006;108:494.
8. Shroyer KR, et al. Validation of a novel immunocytochemical assay for topoisomerase II-a and minichromosome maintenance protein 2 expression in cervical cytology. Cancer. 2006;108:324.
9. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
10. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory Workers from Occupationally Acquired Infections; Approved Guideline-Fourth Edition CLSI document M29-A4 Wayne, PA 2014.