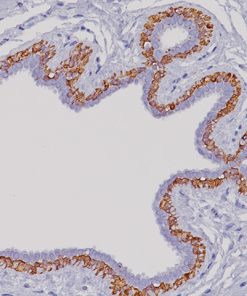
HPV-16
$1,073.00
Description
Product Description
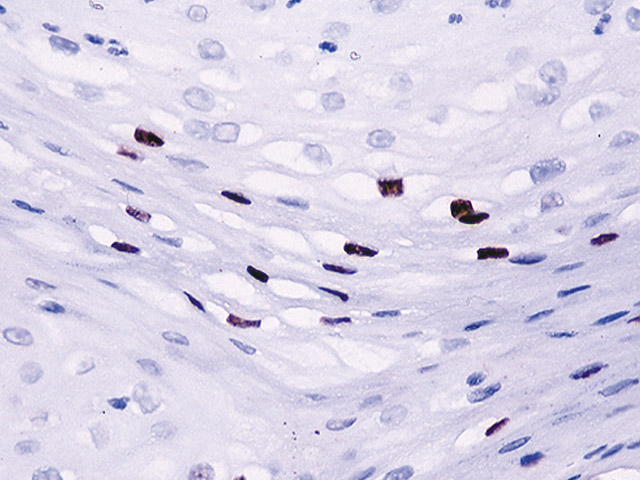
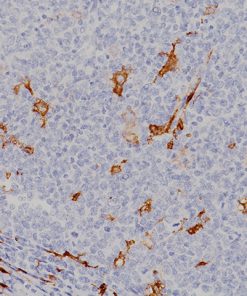
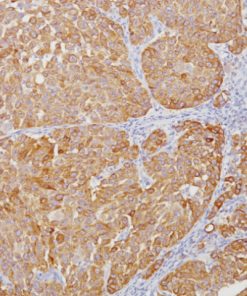
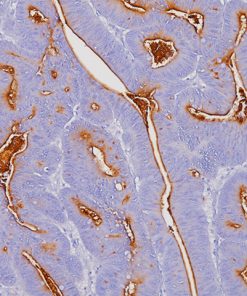
The CAMVIR-1 antibody was raised against the major capsid protein L1 of human papillomavirus HPV-16 antibody, using a recombinant vaccinia virus that expresses the L1 protein. This antibody also detects the HPV-16 L1 antigen in formalin-fixed, paraffin embedded biopsy specimens and on routine cervical smears. The antibody reacts strongly and consistently with specimens containing HPV-16 or HPV-33, but very weak reactions were occasionally observed with biopsy specimens or smears containing HPV-6 or HPV-11.
Specifications
Specifications
| Intended Use | |
|---|---|
| Species Reactivity | |
| Source | |
| Clone | |
| Isotype | |
| Antigen | |
| Localization | |
| Positive Control |
Datasheets & SDS
| Download Data Sheet |
| Download SDS Sheet |
Browse more documents for this product (IFUs, datasheets, translations, SDS, and more).
OUS ONLY IVD
| Download OUS Datasheet |
Legal NoticeIVD is NOT for Sale in the United States but may be available in certain countries outside of the United States, please contact your local distributor for more information. |
References
1. McLean CS, Churcher MJ, Meinke J, Smith GL, Higgins G, Stanley M, Minson AC. Production and characterisation of a monoclonal antibody to human papillomavirus type 16 using recombinant vaccinia virus. J Clin Pathol 1990 Jun;43 (6):488-92.
2. Cowsert LM, Pilacinski WP, Jenson AB. Identification of the bovine papillomavirus L1 gene product using monoclonal Antibodies. Virology 1988 Aug;165(2):613
3. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
4. National Committee for Clinical Laboratory Standards (NCCLS). Protection of laboratory workers from infectious diseases transmitted by blood and tissue; proposed guideline. Villanova, PA 1991;7(9). Order code M29-P.
Related products
Primary Antibodies
Breast