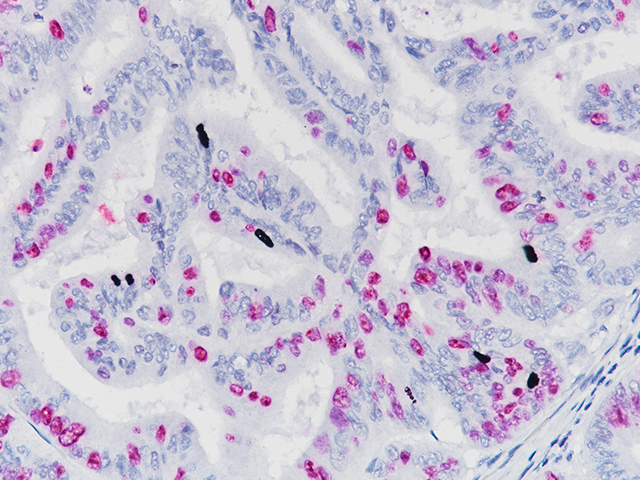
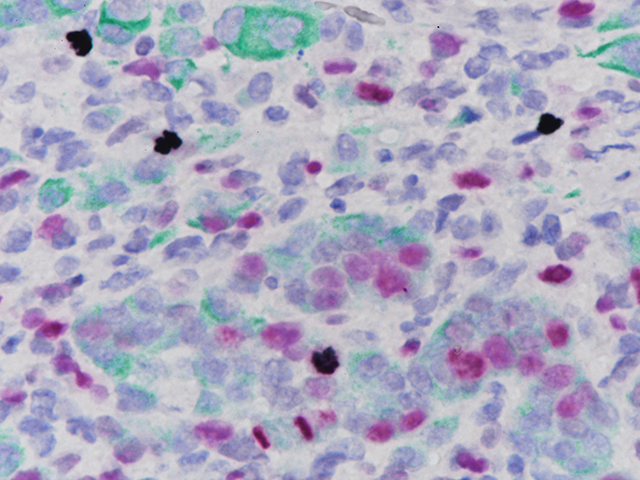
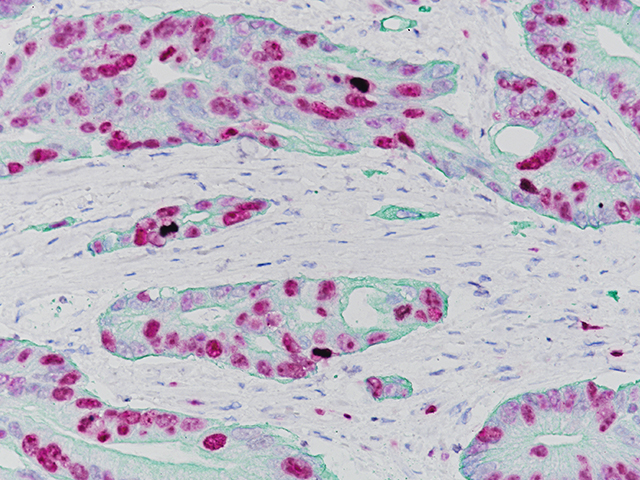
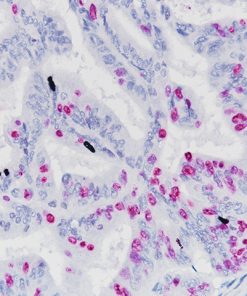
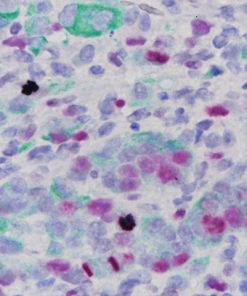
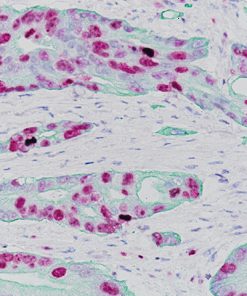
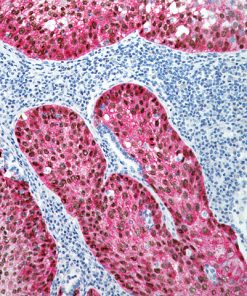
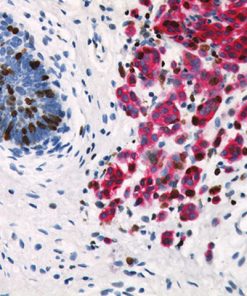
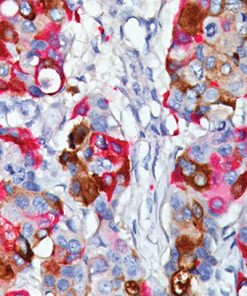
Ki-67 + pHH3
$862.00
Description
Product Description
The Ki-67 nuclear antigen is associated with cell proliferation and found throughout the cell cycle; though not in G0 phase (1,2). The assessment of Ki-67 proliferation in breast cancers has shown the Ki-67 labelling index is an important predictor of survival (3).
Microscopic evaluation of mitotic figures on H&E is a routine procedure in the assessment of the tumor grades (4). However, the counting of mitosis is manual and time consuming with assorted difficulties as well as variabilities between interobserver assessments (5). Histone H3 phosphorylation at Serine10 (pHH3) is in association with mitotic chromatin condensation in late G2 and M phase of the cell cycle. pHH3 can distinguish mitosis from apoptotic nuclei (6). The immunohistochemical staining of Serine10-pHH3 has been reported to be comparable to mitotic figures in the H&E section (7-10).
Immunohistochemical studies have shown immunostaining using anti- Ki-67 and anti-pHH3 antibodies provided improved prediction of recurrence of tumors and may become effective ancillary tools in deciding on optimal therapeutic management (11). Other studies have shown Ki-67 and pHH3 can be useful in separating malignant melanoma from benign nevi (12).
Specifications
Specifications
| Format | |
|---|---|
| Volume | |
| Intended Use | |
| By Letter | |
| Clone | |
| Source |
Datasheets & SDS
References
1. Gerdes J, et al. Cell cycle analysis of a cell proliferation-associated human nuclear antigen defined by the monoclonal antibody Ki-67. J Immunol. 1984 Oct;133(4):1710-5.
2. McCormick D, et al. Detection of the Ki-67 antigen in fixed and waxembedded sections with the monoclonal antibody MIB1. Histopathology. 1993 Apr;22(4):355-60.
3. Pinder SE, et al. Assessment of the new proliferation marker MIB1 in breast carcinoma using image analysis: associations with other prognostic factors and survival. Br J Cancer. 1995 Jan;71(1):146-9.
4. Jannink I, et al. Comparison of the prognostic value of four methods to assess mitotic activity in 186 invasive breast cancer patients: classical and random mitotic activity assessments with correction for volume percentage of epithelium. Hum Pathol. 1995 Oct;26(10):1086-92.
5. Yadav KS, et al. Assessment of interobserver variability in mitotic figure counting in different histological grades of oral squamous cell carcinoma. J Contemp Dent Pract. 2012 May 1;13(3):339-44.
6. Ladstein RG, et al. Prognostic importance of the mitotic marker phosphohistone H3 in cutaneous nodular melanoma. J Invest Dermatol. 2012 Apr;132(4):1247-52.
7. Thareja S, et al. Analysis of tumor mitotic rate in thin metastatic melanomas compared with thin melanomas without metastasis using both the hematoxylin and eosin and anti phosphohistone 3 IHC stain. Am J Dermatopathol. 2014 Jan;36(1):64-7.
8. Ikenberg K, et al. Immunohistochemical dual staining as an adjunct in assessment of mitotic activity in melanoma. J Cutan Pathol. 2012 Mar;39(3):324-30.
9. Casper DJ, et al. Use of anti-phosphohistone H3 immunohistochemistry to determine mitotic rate in thin melanoma. Am J Dermatopathol. 2010 Oct;32(7):650-4.
10. Fulton R, MD, PhD, Tacha D, PhD. Use of a Novel Rabbit Monoclonal Phospho-Histone H3 (Ser10) versus H&E Mitotic Count in Melanoma. USCAP 2016, Poster Session IV #96.
11. Uguen A. et al. Immunostaining of phospho-histone H3 and Ki-67 improves reproducibility of recurrence risk assessment of gastrointestinal stromal tumors. Virchows Arch. 2015 Jul;467(1):47-54.
12. Nasr MR, El-Zammar. Comparison of pHH3, Ki-67, and survivin immunoreactivity in benign and malignant melanocytic lesions. Am J Dermatopathol. 2008 Apr;30(2):117-22.
13. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
14. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory Workers from Occupationally Acquired Infections; Approved Guideline-Fourth Edition CLSI document M29-A4 Wayne, PA 2014.
Related products
Lung
Skin
intelliPATH Antibodies
Breast