Galectin-3
$252.00 – $589.00Price range: $252.00 through $589.00
Description
Product Description
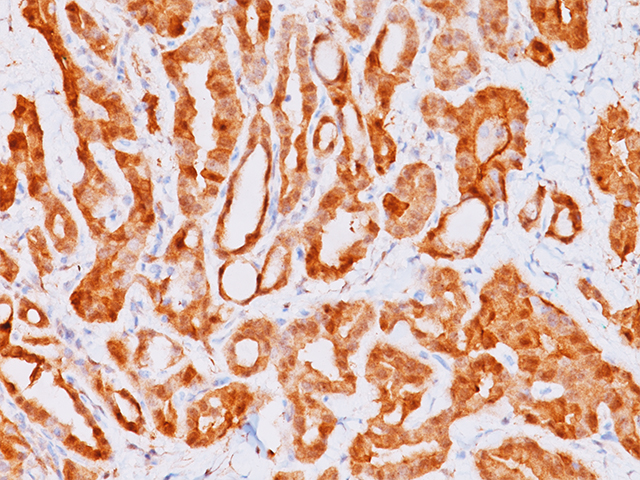
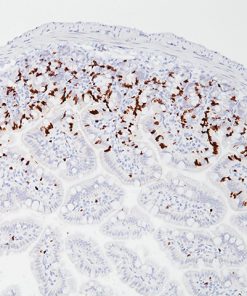
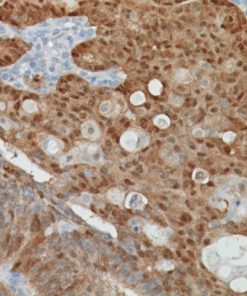
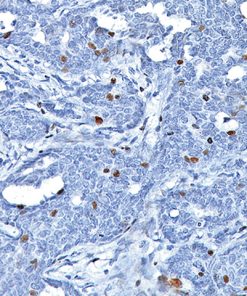
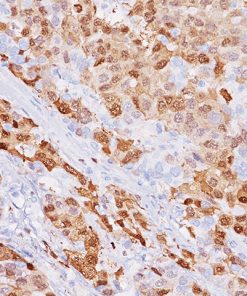
Galectin-3 shows strong diffuse cytoplasmic staining in most cases of papillary thyroid carcinoma (PTC), including the classical and follicular variant (1). Follicular carcinomas with minimal and extensive invasion may also express galectin-3 (2). Most poorly differentiated thyroid cancers demonstrate galectin-3 protein expression while it is not expressed in anaplastic thyroid cancers (3,4). Medullary thyroid carcinomas have shown variable expression of galectin-3 in approximately 50% of cases (4). Galectin-3 is less often detected in follicular thyroid adenomas (0%–37.5%; mean, 21%) (5). Hyperplastic nodules, nodular goiters, and normal follicular epithelium usually show absence of galectin-3 (1-5). Expression of galectin-3 may aid to differentiate malignant from benign thyroid tumors, helping to diagnosis thyroid neoplasms with equivocal morphological features (4-5).
Specifications
Specifications
| By Letter | |
|---|---|
| Antigen | |
| Clone | |
| Format | |
| Intended Use | |
| Isotype | |
| Localization | |
| Positive Control | |
| Source | |
| Species Reactivity | |
| Volume |
Datasheets & SDS
| Download Data Sheet |
| Download SDS Sheet |
Browse more documents for this product (IFUs, datasheets, translations, SDS, and more).
References
1. histomorphologic growth pattern, lymph node metastasis, extrathyroid invasion, and tumor size. Head Neck. 2005 Dec; 27(12):1049-55.
2. Papotti M, et al. Galectin-3 and HBME-1 expression in well-differentiated thyroid tumors with follicular architecture of uncertain malignant potential. Mod Pathol. 2005 Apr; 18(4):541-6.
3. Rossi ED, et al. Diagnostic and prognostic role of HBME-1, Galectin-3, and β- Catenin in poorly differentiated and anaplastic thyroid carcinomas. Appl Immunohistochem Mol Morphol 2013 May; 21(3):237–41.
4. Sumana BS, et al. Galectin-3 immunohistochemical expression in thyroid neoplasms. J Clin Diag Res. 2015 Nov; 9(11): ec07-ec11.
5. Bartolazzi A, et al. Application of an immunodiagnostic method for improving preoperative diagnosis of nodular thyroid lesions. Lancet. 2001 May 26; 357 (9269):1644-50.
6. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
7. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory Workers from Occupationally Acquired Infections; Approved Guideline-Fourth Edition CLSI document M29-A4 Wayne, PA 2014.