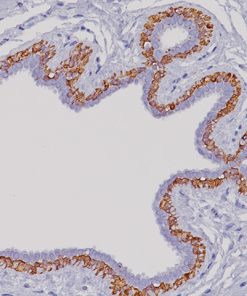
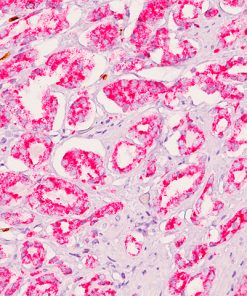
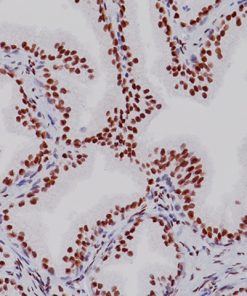
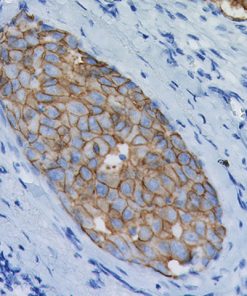
Progesterone Receptor (PR) [16]
$295.00 – $1,308.00Price range: $295.00 through $1,308.00
Description
Product Description
PGR clone 16 is directed against the human progesterone receptor molecule.5-9 PGR clone 16 reacts with both A and B forms of human progesterone receptor by Western blotting procedure.8
Specifications
Specifications
| Intended Use | |
|---|---|
| Species Reactivity | |
| Source | |
| Clone | |
| Isotype | |
| Antigen | |
| Localization | |
| Positive Control |
Datasheets & SDS
| Download ASR Datasheet |
| Download OUS (Outside of the United States) IVD CE Datasheet |
| Download RUO Datasheet |
| Download SDS Sheet |
Browse more documents for this product (IFUs, datasheets, translations, SDS, and more).
References
1. Qiu J, et al. Effect of delayed formalin fixation on estrogen and progesterone receptors in breast cancer: a study of three different clones. Am J Clin Pathol. 2010 Nov; 134(5):813-9.
2. Arihito K, et al. Comparison of evaluations for hormone receptors in breast carcinomas using two manual and three automated immunohistochemical assays. Am J Clin Pathol. 2007 Mar; 127(3):356-65.
3. Press M, et al. Comparison of different antibodies for detection of progesterone receptor in breast cancer steroids. Steroids. 2002 Aug; 67(9):799-813.
4. Mote P, et al. Detection of progesterone receptor forms A and B by immunohistochemical analysis. J Clin Pathol. 2001 Aug; 54(8):624-30.
5. Bevitt D, et al. New monoclonal antibodies to oestrogen and progesterone receptors effective for paraffin section immunohistochemistry. J Pathol. 1997 Oct; 183(3):228-32.
6. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
7. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory workers from occupationally Acquired Infections; Approved guideline-Third Edition CLSI document M29-A3 Wayne, PA 2005.
Related products
Breast
25ml
Breast


![Breast cancer stained with Progesterone Receptor antibody (PR) [16] Breast cancer stained with Progesterone Receptor antibody (PR) [16]](https://biocare.net/wp-content/uploads/sl.jpg)