Description
Product Description
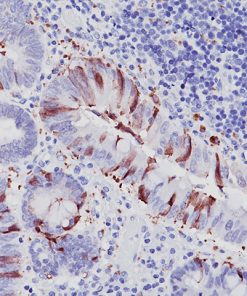
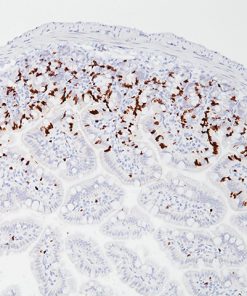
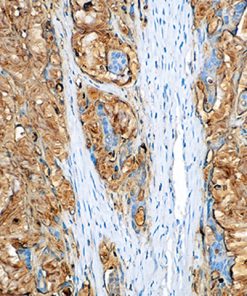
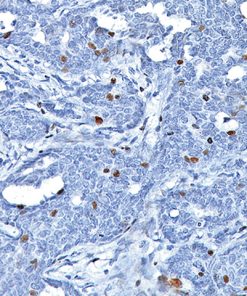
Cytotoxic lymphocytes, including NK cells and cytotoxic T lymphocytes (CTLs), play a major role in the defense against neoplastic processes and viral infections (1). Granule exocytosis is the mechanism by which cytotoxic lymphocytes may induce lysis of its target. This involves granule-associated cytotoxic proteins, including the T-cell intracellular antigen-1 (TIA-1), perforin and granzyme B (1,2). Perforin produces pores that allow the entry of other cytotoxic proteins such as TIA-1 and granzymes, which trigger a process leading to DNA fragmentation and apoptosis of the target cells (1). However, there is evidence that granzyme B may kill targets independently of perforin (2). Recent studies (3,4,5) have demonstrated that the expression of granzyme B by immunohistochemistry in several entities of extranodal peripheral T cell lymphoma (PTCL) and NK cell lymphomas, including nasal and nasal-type NK/T cell lymphomas, hepatosplenic and non-hepatosplenic PTCL, enteropathy-type (ETCL) and non-ETCL intestinal PTCL, subcutaneous panniculitis-like PTCL (SPTCL), cutaneous CD8+ epidermotropic lymphomas as well as in nodal and cutaneous CD30+ anaplastic large cell lymphomas (ALCL) (3,4,5). In contrast, only a few nodal PTCL-unspecified (UC) express cytotoxic phenotype while angioimmunoblastic (AILD) lymphomas do not (3,4,5). The expression of cytotoxic proteins, such as granzyme B may be important for the identification and classification of extranodal T- and NK-cell lymphomas since many of these tumors do not have specific morphology and phenotype (6,7).
Specifications
Specifications
| Format | |
|---|---|
| Volume | |
| Species Reactivity | |
| Clone | |
| Intended Use | |
| Localization | |
| Antigen | Recombinant protein corresponding to the Nterminus of granzyme B |
| Isotype |
Datasheet & SDS
| Download Data Sheet |
| Download SDS Sheet |
Browse more documents for this product (IFUs, datasheets, translations, SDS, and more).
References
1. Smyth MJ, Trapani JA. Granzymes: exogenous proteinases that induce target cell apoptosis. lmmunol Today. 1995 Apr;16(4):202-6.
2. Froelich CJ, Dixit VM, Yang X. Lymphocyte granule-mediated apoptosis: matters of viral mimicry and deadly proteases. Immunol Today. 1998 Jan;19(1):30-6.
3. Krenacs L, et al. Cytotoxic cell antigen expression in anaplastic large cell lymphomas of T- and null-cell type and Hodgkin’s disease: evidence for distinct cellular origin. Blood. 1997 Feb 1;89(3):980-9.
4. Kanavaros P, et al. Cytotoxic protein expression in non-Hodgkin’s lymphoma and Hodgkin’s disease. Anticancer Res. 1999 Mar- Apr;19(2A):1209-16.
5. Kanavaros P, et al. Expression of cytotoxic proteins in peripheral Tcell and natural killer-cell (NK) lymphomas: association with extranodal site, NK or Tgammadelta phenotype, anaplastic morphology and CD30 expression. Leuk Lymphoma. 2000 Jul;38(3-4):317-26.
6. Jaffe ES, et al. Extranodal peripheral T-cell and NK-cell neoplasms. Am J Clin Pathol. 1999 Jan;111(1 Suppl 1):S46-55. Review.
7. Kinney MC. The role of morphologic features, phenotype, genotype and anatomic site in defining extranodal T-cell and NK-cell neoplasms. Am J Clin Pathol. 1999 Jan;111(1 Suppl 1):S104-18.
8. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
9. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory Workers from Occupationally Acquired Infections; Approved Guideline-Fourth Edition CLSI document M29-A4 Wayne, PA 2014.
Related products
Biocare Clone