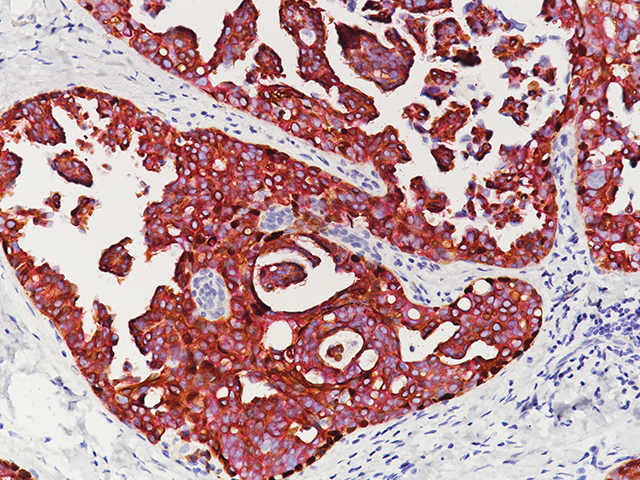
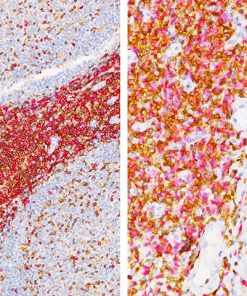
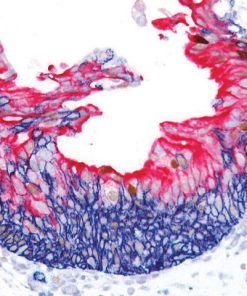
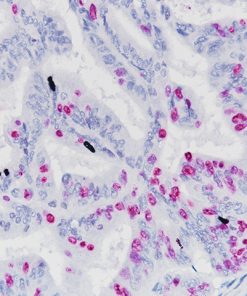
Breast Cocktail (CK HMW/p63 + CK7/8/18)
$1,357.00 – $5,382.00Price range: $1,357.00 through $5,382.00
Description
Product Description
Breast Cocktail (CK HMW/p63 + CK7/8/18) is comprised of mouse monoclonal anti-CK HMW and anti-p63 antibodies as well as rabbit monoclonal anti-CK7 and mouse monoclonal anti-CK8/18 antibodies. CK HMW (high molecular weight cytokeratin) is expressed in the cytoplasm of basal cells and myoepithelium of breast tissue (1-4). p63 is a transcription factor present in the nuclei of myoepithelial cells (2,4). In contrast, CK7, CK8 and CK18 are low molecular weight cytokeratins primarily expressed in luminal cells of the breast (1-3).
CK HMW, p63, CK7, CK8 and CK18 have routinely been used as a panel of IHC markers to complement morphological evaluation in the assessment of breast lesions, due to the differential expression of the luminal vs. basal and myoepithelial markers (1-5). Cases of usual ductal hyperplasia (UDH) have been associated with expression of the basal cell markers, intermixed with cells expressing the keratins of luminal cells (1-2, 6-10). Most cases of atypical ductal hyperplasia (ADH) and low grade ductal carcinoma in situ (LG-DCIS) were negative for the basal markers and exhibited an immunophenotype indicative of luminal cells (1,5-8). Additionally, the basal phenotype has been shown to be characterized by luminal expression of the basal and myoepithelial markers, using a cocktail of CK HMW and p63 (11-13).
IHC, using CK HMW, p63, CK7, CK8 and CK18 antibodies, evaluated in combination with hematoxylin and eosin (H&E), has been shown to significantly increase inter-observer agreement amongst pathologists, compared to H&E alone (14).
Datasheets & SDS
| Download Data Sheet |
| Download RUO Data Sheet |
| Download SDS Sheet |
Browse more documents for this product (IFUs, datasheets, translations, SDS, and more).
References
1. Hicks DG. Immunohistochemistry in the diagnostic evaluation of breast lesions. Appl Immunohistochem Mol Morph. 2011; 19:501-5.
2. Moriya T, et al. New trends of immunohistochemistry for making differential diagnosis of breast lesions. Med Mol Morphol. 2006; 39:8- 13.
3. Yeh IT, Mies C. Application of immunohistochemistry to breast lesions. Arch Pathol Lab Med. 2008; 132:349-57.
4. Lerwill MF. Current practical applications of diagnostic immunohistochemistry in breast pathology. Am J Surg Pathol. 2004; 28:1076-91.
5. Moriya T, et al. Usefulness of immunohistochemistry for differential diagnosis between benign and malignant breast lesions. Breast Cancer. 2009; 16:173-8.
6. Otterbach F, et al. Cytokeratin 5/6 immunohistochemistry assists in differential diagnosis of atypical proliferations of the breast. Histopathology. 2000; 37:232-40.
7. Lacroix-Triki M, et al. Value of cytokeratin 5/6 immunostaining using D5/16 B4 antibody in the spectrum of proliferative intraepithelial lesions of the breast. A comparative study with 34betaE12 antibody. Virchows Arch. 2003; 442:548-54.
8. Boecker W, et al. Usual ductal hyperplasia of the breast is a committed stem (progenitor) cell lesion distinct from atypical ductal hyperplasia and ductal carcinoma in situ. J Pathol. 2002; 198:458-67.
9. Koo JS, et al. Comparison of Immunohistochemical staining in breast papillary neoplasm of cytokeratin 5/6 and p63 in core needle biopsies and surgical excisions. Appl Immunohistochem Mol Morph. 2012; 20:108-15.
10. Ichihara S, et al. Double immunostaining with p63 and highmolecular- weight cytokeratins distinguishes borderline papillary lesions of the breast. Path Int. 2007; 57:126-32.
11. Livasy CA, et al. Phenotypic evaluation of the basal-like subtype of invasive breast carcinoma. Mod Pathol. 2006; 19:264-71.
12. Laakso M, et al. Cytokeratin 5/14-positive breast cancer: true basal phenotype confined to BRCA1 tumors. Mod Pathol. 2005; 18:1321-8.
13. Bhargava R, et al. CK5 is more sensitive than CK5/6 in identifying the “basal-like” phenotype of breast carcinoma. Am J Clin Pathol. 2008; 130:724-30.
14. Jain RK, et al. Atypical ductal hyperplasia: interobserver and intraobserver variability. Mod Pathol. 2011; 24:917-23.
15. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
16. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory Workers from Occupationally Acquired Infections; Approved Guideline-Fourth Edition CLSI document M29-A4 Wayne, PA 2014.
Related products
intelliPATH Antibodies
Biocare Clone