PD-L1
$295.00 – $1,765.00Price range: $295.00 through $1,765.00
Description
Product Description
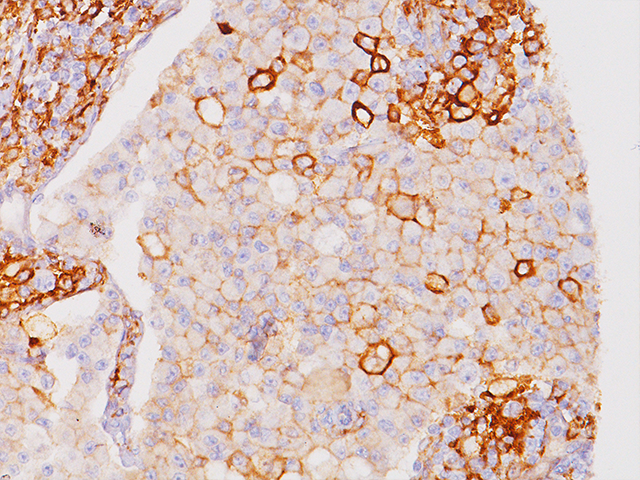
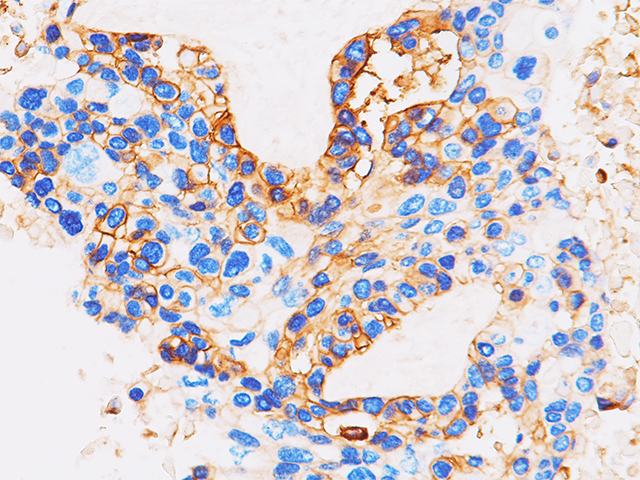
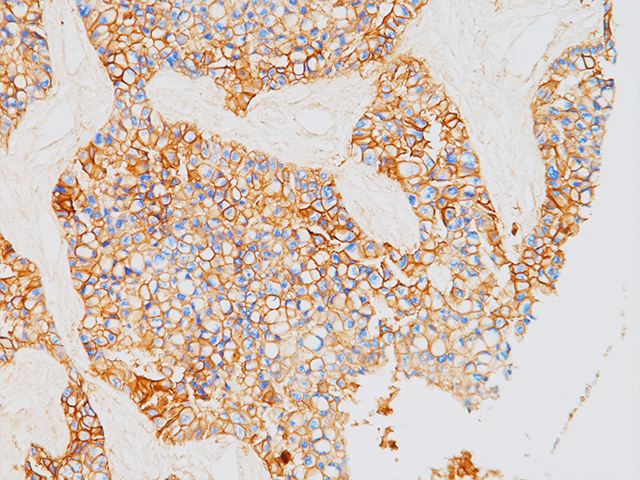
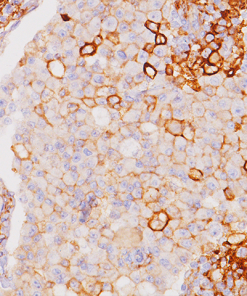
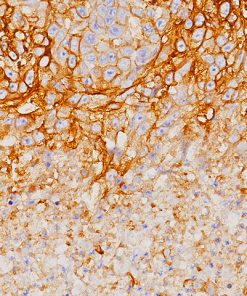
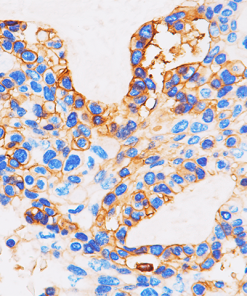
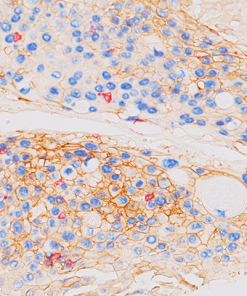
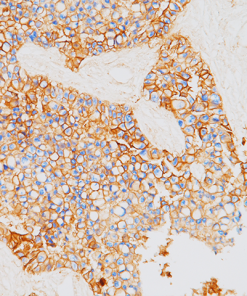
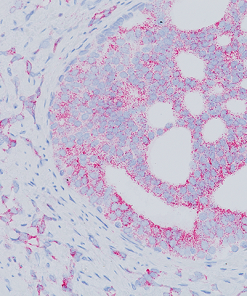
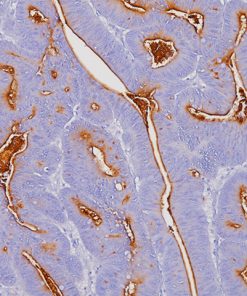
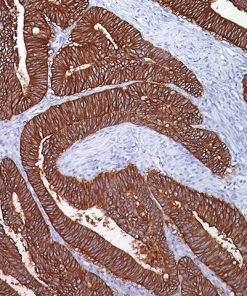
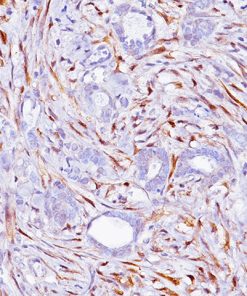
Tumor cells can express the PD-L1 protein to protect themselves from immune system attack by binding to the PD-1 receptor on T cells, which suppresses the T cells’ ability to destroy the tumor.15 PD-L1 is frequently over expressed in placenta, and many human tumors such as melanoma, diffuse large B-cell lymphoma, and carcinomas of the lung, bladder, breast, kidney and colon.16- 18
Specifications
Specifications
| Intended Use | |
|---|---|
| Format | Concentrate, NeoPATH Pro, ONCORE Pro, Predilute, Q Series, UltraLine |
| Volume | |
| Source | |
| Clone | |
| Isotype | |
| Localization | |
| Positive Control |
Datasheets & SDS
| Download CE Marked IVD Datasheet |
| Download IVD Datasheet Without CE Mark |
| Download RUO Data Sheet |
| Download SDS Sheet |
Browse more documents for this product (IFUs, datasheets, translations, SDS, and more).
References
References:
1. Ostrand-Rosenberg S, Horn LA, Haile ST. The programmed death-1 immunesuppressive pathway: barrier to antitumor immunity. J Immunol. 2014 Oct 15;193
(8):3835-41.
2. Tokito T, et al. Predictive relevance of PD-L1 expression combined with CD8+ TIL density in stage III non-small cell lung cancer patients receiving concurrent
chemoradiotherapy. Eur J Cancer. 2016 Jan 6;55:7-14.
3. Park IH, et al. Prognostic implications of tumor-infiltrating lymphocytes in association with programmed death ligand 1 expression in early-stage breast cancer. Clin Breast Cancer. 2016 Feb;16(1):51-8.
4. Kakavand H, et al. Tumor PD-L1 expression, immune cell correlates and PD-1+ lymphocytes in sentinel lymph node melanoma metastases. Mod Pathol. 2015 Dec;28
(12):1535-44.
5. Xia B, Herbst RS. Immune checkpoint therapy for non-small-cell lung cancer: an update. Immunotherapy. 2016 Feb 9 [Epub ahead of print].
6. Patel SP, Kurzrock R. PD-L1 expression as a predictive biomarker in cancer immunotherapy. Mol Cancer Ther. 2015 Apr;14(4):847-56.
7. Singh BP, Salama AK. Updates in therapy for advanced melanoma. Cancers (Basel). 2016 Jan 15;8(1).
8. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
9. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory Workers from Occupationally Acquired Infections; Approved Guideline-Fourth Edition CLSI document M29-A4 Wayne, PA 2014.