Fixated on the Basics
The success of an immunohistochemical (IHC) process can be linked to pre-analytical steps of the histological process, as these steps can
significantly and adversely affect accurate detection of targets. Preserving the integrity of tissue collected for pathological examination is of
utmost importance and is achieved through a process called fixation. While chemical fixation denatures and stabilizes proteins, it can also
destroy and mask antigenic targets.1
When preparing tissue sections for IHC, fixation composition should be carefully considered. Each fixative has its own unique preservation
characteristics and can impact variables such as antigen retrieval and antibody binding. While scientists have formulated many different
fixative solutions, most fixatives can be divided into 3 categories: formalin-based, alcohol-based, and solutions containing of both formalin
and alcohol. There is not one single fixative that is ideal for all antigens; however, the most common fixative in pathology is 10% neutral
buffered formalin (NBF) with a pH of 7.0-7.4.1 NBF fixes tissue by penetrating and forming cross linkages between reactive amino groups in
proteins.2 Due to this crosslinking, proteins, or epitopes, usually require an “unmasking” process or “antigen retrieval” step.1,2 Various antigen
retrieval techniques have been developed to enable target antibodies to bind to their corresponding antigen site after NBF fixation. For more
information on antigen retrieval, please see Biocare’s Whitepapers entitled:
“The Basic Truth: Antigen Retrieval Solutions and pH”
&
“NxGeneration of Offline Antigen Retrieval Devices.”
The duration of time before placement into a fixative and the time spent in a fixative can greatly affect IHC results. Delayed fixation has
shown to cause false negatives, due to cell degradation and loss of target protein structures.1 Under fixation may result in a “fading” of
immunoreactivity toward the center of tissues, as the fixative fails to fully penetrate the tissue.1 Over fixation can result in excessive masking
of the epitope which may be difficult to reverse, leading to weakened immunoreactivity. These effects have led to confusion about what is
an acceptable length of time for NBF to properly fix specimen.1,2 As a general rule of thumb, fixation time for tissue measuring 4 mm thick
is “at least 24 hours at room temperature.”2 To help standardize the grading scale for breast markers, the ASCO/CAP has also created a
recommendation guideline specifically for fixing breast tissue. The guideline states that “10% NBF fixation, prior to IHC labeling, of ER, PgR,
and HER2 cannot be less than 6 hours and should be no more than 72.”1,2
When troubleshooting IHC, it is important to consider all stages: pre-analytical, analytical, and post-analytical. As fixation takes place in the
pre-analytical stage, it could easily be overlooked; however, research proves fixation profoundly affects the immunoreactivity and cellular
preservation of patient tissue.
Having staining issues or need guidance optimizing your antibody protocols? Biocare Medical offers outstanding technical support and field-based
services. Our expert team of highly experienced professionals can provide training and troubleshooting for your Biocare reagents and instrumentation.
Have questions on available services, please contact your local Sales Representative or call 1-800-799-9499.
Effects of Fixation
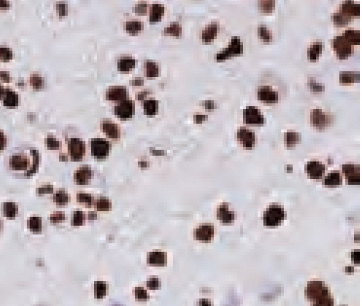
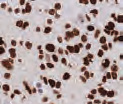
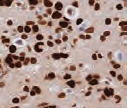
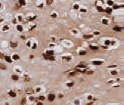
Cell pellets with immediate and delayed fixation times in 10% NBF. Progressive loss of cellular detail and an increase in background (KI-67 staining should be localized only in the nucleus) were exhibited with increased delay of fixation.2
Under fixation resulted in weakening/loss of immunoreactivity toward center of tissue.1
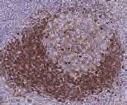
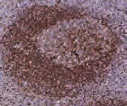
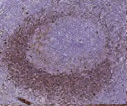

Tonsil with different fixation times- antigen is unmasked at 8 and 32 hours but depletes at 56 and 104 hours, resulting in a loss of reactivity.2
1. Carson, F. L., & Cappellano, C. H. (2015). Histotechnology: A Self Instructional Text (4th ed.). Chicago, Illinois: ASCP Press.
2. Colley, E. C., & Stead, R.H. (2013). Fixation and Other Pre-Analytical Factors. In C. R. Taylor & L. Rudbeck (Eds.), Immunohistochemical Staining Methods (6th ed., pp. 20-29). Dako Denmark A/S.


