Uroplakin II
$238.00 – $2,462.00
Description
Product Description
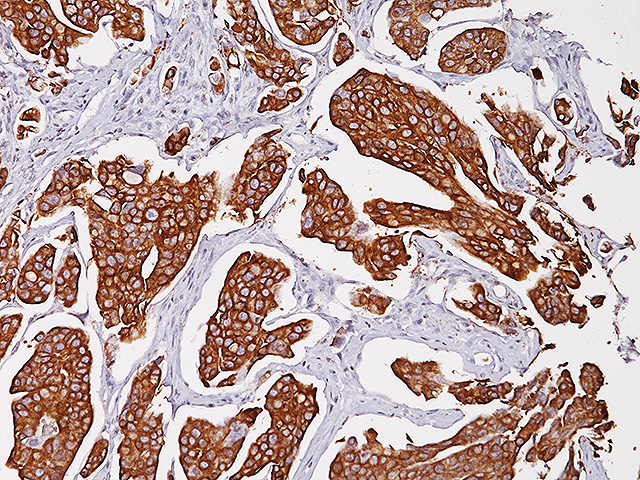
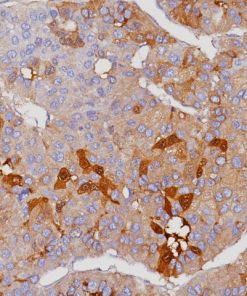
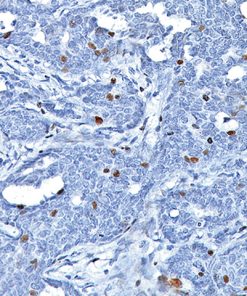
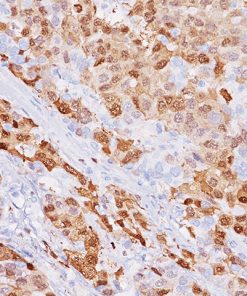
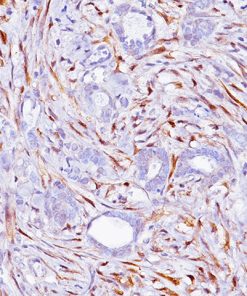
Uroplakin II is a 15 kDa protein component of urothelial plaques. Studies have shown Uroplakin II mRNA was highly specific and was expressed in both bladder cancer tissues and peripheral blood of patients with primary and metastatic urothelial carcinoma of the bladder. Uroplakin II antibody [BC21] has exhibited an increased sensitivity (46/59, 78%) when compared to Uroplakin III [AU1] (191/56, 34%) in cases of urothelial carcinoma of the bladder with the exception of bladder and ureter, staining was highly specific in various normal and neoplastic tissues in an in-house study. Uroplakin II antibody [BC21] is a highly specific antibody that may be useful in identifying tumors of urothelial origin. PATENT PENDING.
Specifications
Specifications
| Weight | N/A |
|---|---|
| Dimensions | N/A |
| Intended Use | |
| Species Reactivity | |
| Source | |
| Clone | |
| Isotype | |
| Antigen | |
| Localization | |
| Positive Control |
Datasheets & SDS
| Download Data Sheet |
| Download Q Series Data Sheet |
| Download RUO Data Sheet |
| Download SDS Sheet |
Regulatory Notice: Biocare’s IVD-labeled products comply with US-FDA and European IVDD regulation. Other regions may have additional requirements for such labeling, please contact your local distributor.
References
1. Wu XR, et al. Uroplakins in urothelial biology, function, and disease. Kidney Int. 2009 Jun; 75(11):1153-65.
2. Wu X, et al. Uroplakin II as a promising marker for molecular diagnosis of nodal metastases from bladder cancer: comparison with cytokeratin 20. J Urol. 2005 Dec; 174(6):2138-42.
3. Lu JJ, et al. Detection of circulating cancer cells by reverse transcription-polymerase chain reaction for uroplakin II in peripheral blood of patients with urothelial cancer. Clin Cancer Res. 2000 Aug; 6(8):3166-71.
4. Li SM, et al. Detection of circulating uroplakin-positive cells in patients with transitional cell carcinoma of the bladder. J Urol. 1999 Sep; 162(3 Pt 1):931-5.
5. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
6. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory workers from occupationally Acquired Infections; Approved guideline-Third Edition CLSI document M29-A3 Wayne, PA 2005.
Reviews (0)
Be the first to review “Uroplakin II” Cancel reply
You must be logged in to post a review.
This site uses Akismet to reduce spam. Learn how your comment data is processed.
Related products
B


Reviews
There are no reviews yet.