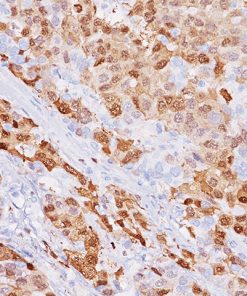
CD33
$298.00 – $1,434.00
Description
Product Description
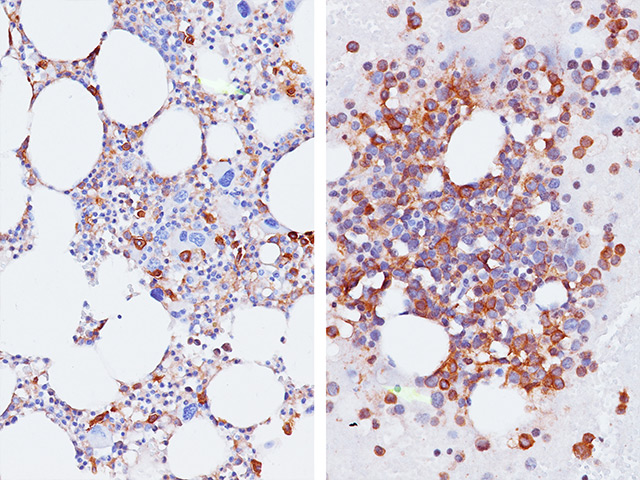
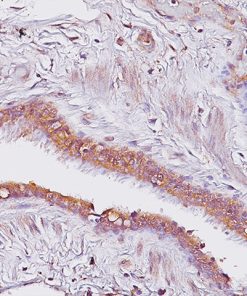
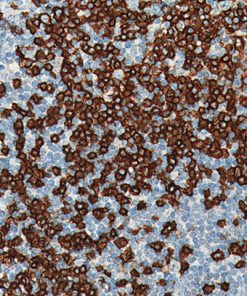
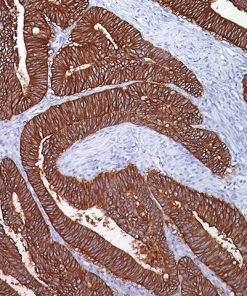
CD33 or Siglec-3 is a 67 kD glycosylated transmembrane receptor expressed on myeloid-specific cells (1,2). In the past, CD33 was only used for flow cell cytometry. Recently, a CD33 for paraffin-embedded tissues has been developed and has been used to phenotype acute myelogenous leukemias (1,2). In cases of acute leukemia, the CD33 antibody showed equivalent results by immunohistochemical analysis compared with flow cytometric analysis (1). The CD33 antibody was also found to be a useful marker in the workup of myeloid sarcomas (1,3). In normal bone marrow trephine biopsies, clone PWS44 stains myeloid, myelomonocytic hemopoiesis and mature macrophages; cells of the erythroid and megakaryocytes series are negative for CD33 (1). In conclusion, CD33 antibody may be a useful marker as part of an antibody panel for the identification of acute leukemias, myeloid proliferative disorders and myeloid sarcomas on paraffin-embedded tissue samples (1-3).
Specifications
Specifications
| Intended Use | |
|---|---|
| Format | |
| Volume | |
| Source | |
| Clone | |
| Isotype | |
| Antigen | Prokaryotic recombinant protein corresponding to a region of the C2 domain on human CD33 |
| Localization | |
| Positive Control |
Datasheets & SDS
References
1. Hoyer JD, et al. CD33 detection by immunohistochemistry in paraffin-embedded tissues: a new antibody shows excellent specificity and sensitivity for cells of myelomonocytic lineage. Am J Clin Pathol. 2008 Feb; 129(2):316-23.
2. Rollins-Raval MA, Roth CG. The value of immunohistochemistry for CD14, CD123, CD33, myeloperoxidase and CD68R in the diagnosis of acute and chronic myelomonocytic leukaemias. Histopathology. 2012 May; 60(6):933-42.
3. Amador-Ortiz C, et al. Use of classic and novel immunohistochemical markers in the diagnosis of cutaneous myeloid sarcoma. J Cutan Pathol. 2011 Dec; 38(12):945-53.
4. Brotelle T, et al. Gemtuzumab ozogamicin for treatment of acute myeloid leukemia. Bull Cancer. 2014 Feb; 101(2):211-8.
5. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
6. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory Workers from Occupationally Acquired Infections; Approved Guideline-Fourth Edition CLSI document M29-A4 Wayne, PA 2014.
Related products
B